Ionis Vs. Covalent Bonds
Kailyn B. / Aiyana H.
Ionic Bonding & Covalent Bonding
AIonic bonding- is the complete transfer of valence electrons between atoms. It is a type of chemical bond that generates two oppositely charged ions. In ionic bond, the metal loses electrons to become a positively charged cation, whereas the non-metal accepts those electrons to become a negatively charged anion
covalent bond -is a chemical bond that involves the sharing of electron pairs between atoms. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding.
example of ionic bond
Na-Cl
Sodium Chloride AKA Table Salt
Sodium Chloride AKA Table Salt
example of covalent bond
H2O
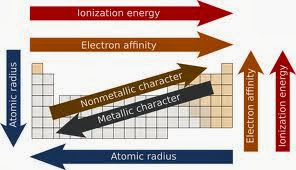
Periodic Trends And How They Affect Bonding
Periodic trends affect bonding, because of how the elements are arranged on the periodic table. For example elements can be arranged by their electronegative, electron affinity, atomic radius, or ionization energy. Electronegative is the atoms ability to attract other bonded atoms. Electron affinity is an atoms ability to attract another atom. The atomic radius is the radius of an elements atom. Ionization energy is the energy it takes to remove an atom from another atom. Other periodic trends are when the attraction of the atoms for the pair of bonding electrons is different, this is polar covalent bonds. Properties in compounds are used to determine the type of bonding and structure, not just the elements being used. These different properties help group elements to make them either more available or less available for bonding.